Thank you for reading this post, don't forget to subscribe!
- Applied Catalysis B: Environmental, Volume 188, 5 July 2016, Pages 217-226
- DOI: https://doi.org/10.1016/j.apcatb.2016.02.003
- Project: Synthesis, Characterization of Hybrid Materials Incuding Water Soluble Metallophthalocyanine Complexes With Functionalized Graphene and Investigation of Physical and Sensor Properties
- Cite this: App. Cat. B Env., 188, 5 July 2016, 217-226.
Abstract and figures:
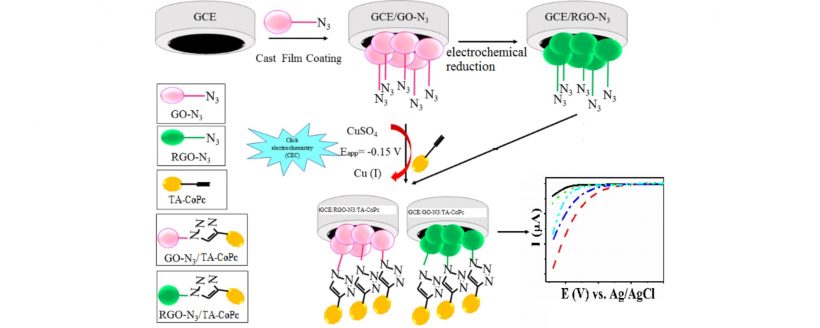
Electrocatalytic hydrogen evolution reaction (HER) on azido graphene oxide (GO-N3) and reduced azido graphene oxide (RGO-N3) electrodes decorated with the cobaltphthalocyanine complex bearing terminal alkyne moieties (TA-CoPc) was investigated. GCE/RGO-N3 electrode was constructed with the electrochemical reduction of GO-N3 coated on a glassy carbon electrode. Decoration of GCE/RGO-N3 and GCE/GO-N3 electrodes were performed with a new electrode modification technique, “click electrochemistry (CEC)”, with which TA-CoPc complex was bonded to azido functional groups of GO-N3 and RGO-N3 on the electrodes. The modified GCE/RGO-N3/TA-CoPc and GCE/GO-N3/TA-CoPc electrodes were characterized with square wave voltammetry and electrochemical impedance spectroscopy (EIS), and then tested as heterogeneous electrocatalysts for HER. GCE/RGO–N3/TA-CoPc electrode illustrates well electrocatalytic activity by decreasing the over-potential of the bare electrode about 340 mV and increasing the current density of the electrode about 15 fold at low pHs with absolutely high stability and reproducibility.
SUBJECT: Electrocatalyst, Hydrogen evolution reaction, Phthalocyanine, Click electrochemistry, Graphene Oxide, Reduced graphene oxide


Fig. 8. CCs of GCE/GO-N3, GCE/RGO-N3, GCE/GO-N3/TA-CoPc, and GCE/RGO-N3/TA-CoPc electrodes recorded at 0.010 mV s−1 scan rate with 1000 rpm rotating speed in PBS containing 0.1 M LiClO4. (a) pH 3.40; (b) pH 5.60; (c) pH 7.40; (d) pH 10.00. (pHs of the electrolyte was adjusted with conjugated acid of the buffer solution).

 Duygu Akyüz
Duygu Akyüz Bahadir Keskin*
Bahadir Keskin* Utkan Şahintürk
Utkan Şahintürk ATIF KOCA
ATIF KOCA





